Introduction to Actin Bundle Assembly[Edit]
Crosslinking of actin filaments is a critical step in cell motility and is a fundamental process in filopodia protrusion and lamellipodia formation.
 Figure 1. Types of actin filament crosslinking proteins: Smaller cross-linking proteins that are more globular (e.g. fascin) or have more than one actin binding site (e.g. fimbrin, α-actinin dimers) primarily form actin bundles. Larger crosslinking proteins (e.g. spectrin, filamin, dystrophin) create more space between actin filaments and they generally form actin networks. Other actin crosslinking proteins not shown here include: scruin, dematin, and villin.In filopodia, crosslinking of actin filaments provides the rigidity needed to overcome the compressive force of the plasma membrane, which individual actin filaments otherwise lack [1, 2]. Filopodia in nerve growth cones contain tightly packed bundles of actin filaments that usually contain more than 15 parallel filaments. These are likely to be oriented with their barbed ends towards the tip [3]. Mechanically, a crosslinked filopodial bundle functions as an effective elastic rod. Bundle stiffness increases with the number of bundled filaments and so contributes to the overall filopodium length [1].
Figure 1. Types of actin filament crosslinking proteins: Smaller cross-linking proteins that are more globular (e.g. fascin) or have more than one actin binding site (e.g. fimbrin, α-actinin dimers) primarily form actin bundles. Larger crosslinking proteins (e.g. spectrin, filamin, dystrophin) create more space between actin filaments and they generally form actin networks. Other actin crosslinking proteins not shown here include: scruin, dematin, and villin.In filopodia, crosslinking of actin filaments provides the rigidity needed to overcome the compressive force of the plasma membrane, which individual actin filaments otherwise lack [1, 2]. Filopodia in nerve growth cones contain tightly packed bundles of actin filaments that usually contain more than 15 parallel filaments. These are likely to be oriented with their barbed ends towards the tip [3]. Mechanically, a crosslinked filopodial bundle functions as an effective elastic rod. Bundle stiffness increases with the number of bundled filaments and so contributes to the overall filopodium length [1].
In lamellipodia, crosslinking also strengthens the actin filaments, however in this case the filaments form a branched network, which is connected at certain points to membrane bound proteins and focal adhesions (as depicted in Fig 1). Crosslinking also increases the ATPase activity of myosins and increases the tension on actin filaments [4].
A number of proteins including fascin, filamin, α-actinin, and members of the I-BAR family of proteins serve as functional modules in actin crosslinking (see figure below). In each case these proteins bind to two actin filaments, and in some cases, additional regulatory proteins. Despite considerable redundancy between F-actin crosslinking proteins, their specific subcellular localization suggests that each of these proteins may play a unique role in coordinating the organization of actin (reviewed in [5]).
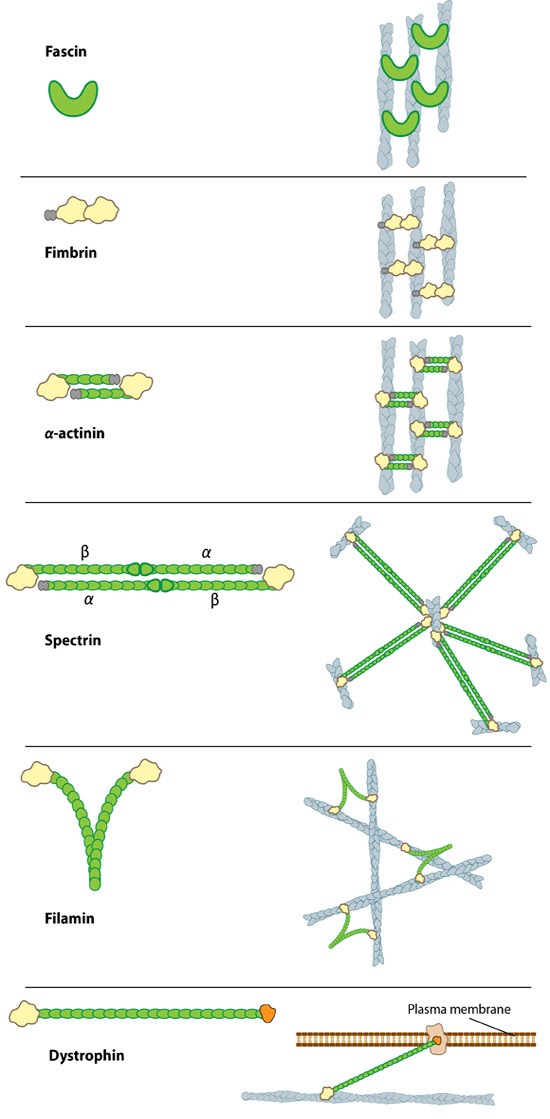 Figure 1. Types of actin filament crosslinking proteins: Smaller cross-linking proteins that are more globular (e.g. fascin) or have more than one actin binding site (e.g. fimbrin, α-actinin dimers) primarily form actin bundles. Larger crosslinking proteins (e.g. spectrin, filamin, dystrophin) create more space between actin filaments and they generally form actin networks. Other actin crosslinking proteins not shown here include: scruin, dematin, and villin.
Figure 1. Types of actin filament crosslinking proteins: Smaller cross-linking proteins that are more globular (e.g. fascin) or have more than one actin binding site (e.g. fimbrin, α-actinin dimers) primarily form actin bundles. Larger crosslinking proteins (e.g. spectrin, filamin, dystrophin) create more space between actin filaments and they generally form actin networks. Other actin crosslinking proteins not shown here include: scruin, dematin, and villin.In lamellipodia, crosslinking also strengthens the actin filaments, however in this case the filaments form a branched network, which is connected at certain points to membrane bound proteins and focal adhesions (as depicted in Fig 1). Crosslinking also increases the ATPase activity of myosins and increases the tension on actin filaments [4].
A number of proteins including fascin, filamin, α-actinin, and members of the I-BAR family of proteins serve as functional modules in actin crosslinking (see figure below). In each case these proteins bind to two actin filaments, and in some cases, additional regulatory proteins. Despite considerable redundancy between F-actin crosslinking proteins, their specific subcellular localization suggests that each of these proteins may play a unique role in coordinating the organization of actin (reviewed in [5]).