Lamellipodium Assembly
Content
Introduction to Lamellipodia Assembly[Edit]
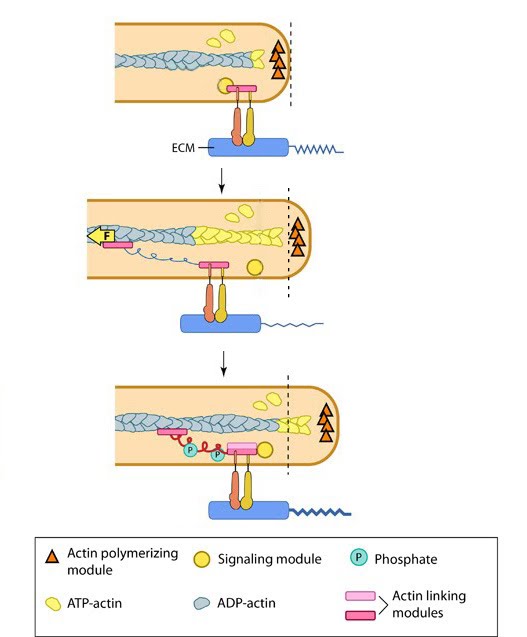
The lamellipodia is a distinct region of the cell that facilitates cell motility and various mechanosensing mechanisms. Lamellipodium assembly can be described in a series of defined steps, many of which involve processes related to cytoskeletal dynamics, and which utilize several “functional modules”.
The actin cytoskeleton plays an essential roll in the formation and function of the lamellipodia. Lamellipodial actin filaments are highly dynamic, especially compared to those of the lamella [1] and it is due to their dynamic nature, and the constant cycles of actin filament polymerization and actin filament depolymerization that the protrusive force required to stretch the membrane and allow the lamellipodia to spread, is generated.
 Figure 1. Conserved steps in cell spreading and movement: 1. Polymerization of actin filaments at the leading edge is translated into protrusive force. 2. Membrane protrusion facilitates the binding of transmembrane cell surface receptors to the substratum components. New adhesions are rapidly linked to the network of actin filaments. 3. The combined activity of retrograde actin movement and contractile forces produced by stress fibers generate tension to pull the cell body forward. 4. The forces produced by the contractile network combined with actin filament and focal adhesion disassembly, helps to retract the trailing cell edge. (Note: the “space” between the cell and the substrate [shown as a gray bar] is exaggerated in this diagram).
Contractions of the actomyosin network are required for both mechanosensing and cell motility mechanisms. In the former scenario, contractions are required for the cell to pull against its substrate, thereby measuring its rigidity. In migrating cells, contractions are required to translate generated forces into motility, pulling the cell against the focal adhesions and facilitating the retraction of the trailing edge. The final step in the function of the lamellipoida/lamella is the retraction of the trailing edge. This ensures the cell is not permanently anchored to its substrate and therefore allows a net gain in forward movement
Figure 1. Conserved steps in cell spreading and movement: 1. Polymerization of actin filaments at the leading edge is translated into protrusive force. 2. Membrane protrusion facilitates the binding of transmembrane cell surface receptors to the substratum components. New adhesions are rapidly linked to the network of actin filaments. 3. The combined activity of retrograde actin movement and contractile forces produced by stress fibers generate tension to pull the cell body forward. 4. The forces produced by the contractile network combined with actin filament and focal adhesion disassembly, helps to retract the trailing cell edge. (Note: the “space” between the cell and the substrate [shown as a gray bar] is exaggerated in this diagram).
Contractions of the actomyosin network are required for both mechanosensing and cell motility mechanisms. In the former scenario, contractions are required for the cell to pull against its substrate, thereby measuring its rigidity. In migrating cells, contractions are required to translate generated forces into motility, pulling the cell against the focal adhesions and facilitating the retraction of the trailing edge. The final step in the function of the lamellipoida/lamella is the retraction of the trailing edge. This ensures the cell is not permanently anchored to its substrate and therefore allows a net gain in forward movement
The above mentioned activities produce net forward/backward cell movement or spreading of the plasma membrane in a specific direction (aka polarized movement). It should be noted however that the specific biophysical and biochemical parameters that are altered at each step have been difficult to measure due to the wide variety of motile structures that can be found in each cell at any given time. For example, migratory fibroblasts exhibit lamellipodia protrusion and retraction, ruffling, filopodial protrusion and retraction, bleb protrusion and retraction, trailing edge retraction, and quiescence in neighboring regions of the cell edge [2].
Fortunately, recent advances in technological systems that measure the temporal and spatial movement of single proteins in specific cell structures, whole cells, and tissues has greatly expanded our mechanistic understanding of cell motility and the composite functional modules [2, 3, 4, 5, 6] (reviewed in [7]). Surprisingly, recent work has suggested that the basic mechanism for polarization and directional movement lies in the microtubules, which can be modified by their interaction with the actin-myosin system and cell-substrate adhesions [8]. Although there are numerous details that remain unresolved, it is abundantly clear that mechanical mechanisms are essential for coordinating the physical and biochemical processes that determine cell shape and locomotion.
The actin cytoskeleton plays an essential roll in the formation and function of the lamellipodia. Lamellipodial actin filaments are highly dynamic, especially compared to those of the lamella [1] and it is due to their dynamic nature, and the constant cycles of actin filament polymerization and actin filament depolymerization that the protrusive force required to stretch the membrane and allow the lamellipodia to spread, is generated.
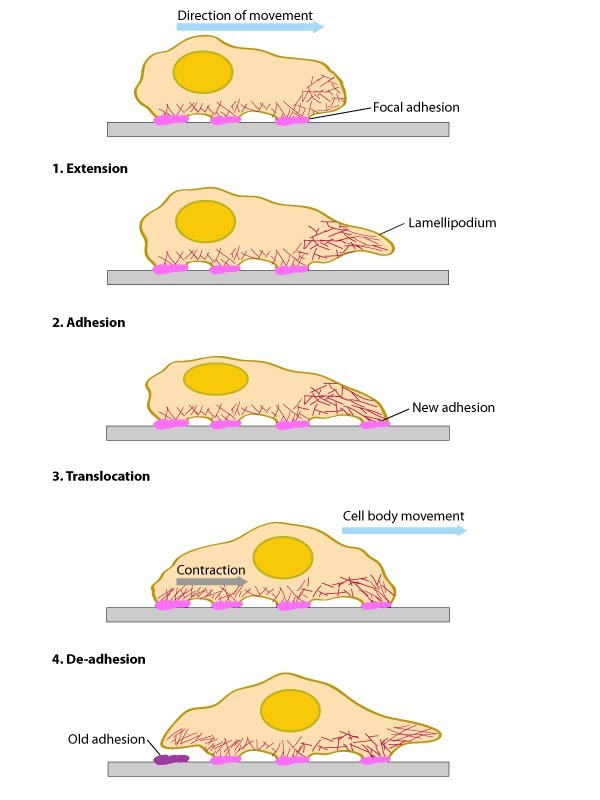 Figure 1. Conserved steps in cell spreading and movement: 1. Polymerization of actin filaments at the leading edge is translated into protrusive force. 2. Membrane protrusion facilitates the binding of transmembrane cell surface receptors to the substratum components. New adhesions are rapidly linked to the network of actin filaments. 3. The combined activity of retrograde actin movement and contractile forces produced by stress fibers generate tension to pull the cell body forward. 4. The forces produced by the contractile network combined with actin filament and focal adhesion disassembly, helps to retract the trailing cell edge. (Note: the “space” between the cell and the substrate [shown as a gray bar] is exaggerated in this diagram).
Figure 1. Conserved steps in cell spreading and movement: 1. Polymerization of actin filaments at the leading edge is translated into protrusive force. 2. Membrane protrusion facilitates the binding of transmembrane cell surface receptors to the substratum components. New adhesions are rapidly linked to the network of actin filaments. 3. The combined activity of retrograde actin movement and contractile forces produced by stress fibers generate tension to pull the cell body forward. 4. The forces produced by the contractile network combined with actin filament and focal adhesion disassembly, helps to retract the trailing cell edge. (Note: the “space” between the cell and the substrate [shown as a gray bar] is exaggerated in this diagram).The above mentioned activities produce net forward/backward cell movement or spreading of the plasma membrane in a specific direction (aka polarized movement). It should be noted however that the specific biophysical and biochemical parameters that are altered at each step have been difficult to measure due to the wide variety of motile structures that can be found in each cell at any given time. For example, migratory fibroblasts exhibit lamellipodia protrusion and retraction, ruffling, filopodial protrusion and retraction, bleb protrusion and retraction, trailing edge retraction, and quiescence in neighboring regions of the cell edge [2].
Fortunately, recent advances in technological systems that measure the temporal and spatial movement of single proteins in specific cell structures, whole cells, and tissues has greatly expanded our mechanistic understanding of cell motility and the composite functional modules [2, 3, 4, 5, 6] (reviewed in [7]). Surprisingly, recent work has suggested that the basic mechanism for polarization and directional movement lies in the microtubules, which can be modified by their interaction with the actin-myosin system and cell-substrate adhesions [8]. Although there are numerous details that remain unresolved, it is abundantly clear that mechanical mechanisms are essential for coordinating the physical and biochemical processes that determine cell shape and locomotion.
Nucleation of Actin initiates lamellipodia formation[Edit]
In the first phase of lamellipodia formation, actin filament polymerization produces a protrusive force on the cell membrane that promotes the spreading out and enlargement of the lamellipodia. In polarized, migrating cells this is known as the leading edge.
Actin polymerization begins with nucleation of actin filaments, and in the lamellipodia, this is primarily mediated by the Arp2/3 complex.This process, which has become known as the ‘dendritic nucleation model’ [9] is widely accepted despite several papers bringing this model, which is also known as the ‘branched model’ into question in recent years. For example, one study reported a lack of branched filaments in lamellipodia of 3T3 fibroblasts and another study suggested branches are an experimental artifact [10]. Subsequent re-analysis of results reported in this study, which suggested a predominantly unbranched array, revealed the existence of over 200 branched filaments [11].
Current evidence suggests multiple nucleators may function alongside the Arp2/3 complex [1], although the extent to which they influence the growth of the actin filament network and its ability to exert a protrusive force on the cell membrane remains unclear. For example, members of the formin family of nucleators have been found to localize to lamellipodia and were implicated in the nucleation of unbranched filaments [12]. Similar reports suggest spire and other proteins possessing multiple WH2 domains (e.g. JMY [13]; cordon bleu [14]) are involved in unbranched actin filament nucleation within lamellipodia. WH2 (Wiskott-Aldrich homology 2) domains are highly evolutionarily conserved domains of approximately 35 amino acids in length and serve as actin binding sites [15]. A mechanism by which WH2 domain-containing proteins promote filament nucleation has been proposed [16], however recent studies have indicated a greater level of complexity surrounds the biological function of the WH2 domains. For example, it has been shown that Cordon bleu functions with similar characteristics to profilin, being a weak nucleator, but also readily severs filaments and sequesters ADP-actin [17]. Nevertheless, proteins possessing WH2 domains are likely to have an influence on actin filament nucleation in lamellipodia, albeit at a regulatory level or supplementary to the influence of Arp2/3 itself.
There is indirect evidence suggesting that actin may exist in an oligomer state, neither as F-actin nor G-actin, yet diffuses like G-actin, except with slower dynamics. Fast turnover rates for F-actin in lamellipodia are challenging to explain with existing models of actin treadmilling, and some believe that oligomeric actin fragments disassemble from the barbed ends of F-actin [18]. Indirect evidence suggests that actin oligomers could re-associate with the F-actin network, or break up into G-actin monomers, after a characteristic time [19].
Current evidence suggests multiple nucleators may function alongside the Arp2/3 complex [1], although the extent to which they influence the growth of the actin filament network and its ability to exert a protrusive force on the cell membrane remains unclear. For example, members of the formin family of nucleators have been found to localize to lamellipodia and were implicated in the nucleation of unbranched filaments [12]. Similar reports suggest spire and other proteins possessing multiple WH2 domains (e.g. JMY [13]; cordon bleu [14]) are involved in unbranched actin filament nucleation within lamellipodia. WH2 (Wiskott-Aldrich homology 2) domains are highly evolutionarily conserved domains of approximately 35 amino acids in length and serve as actin binding sites [15]. A mechanism by which WH2 domain-containing proteins promote filament nucleation has been proposed [16], however recent studies have indicated a greater level of complexity surrounds the biological function of the WH2 domains. For example, it has been shown that Cordon bleu functions with similar characteristics to profilin, being a weak nucleator, but also readily severs filaments and sequesters ADP-actin [17]. Nevertheless, proteins possessing WH2 domains are likely to have an influence on actin filament nucleation in lamellipodia, albeit at a regulatory level or supplementary to the influence of Arp2/3 itself.
There is indirect evidence suggesting that actin may exist in an oligomer state, neither as F-actin nor G-actin, yet diffuses like G-actin, except with slower dynamics. Fast turnover rates for F-actin in lamellipodia are challenging to explain with existing models of actin treadmilling, and some believe that oligomeric actin fragments disassemble from the barbed ends of F-actin [18]. Indirect evidence suggests that actin oligomers could re-associate with the F-actin network, or break up into G-actin monomers, after a characteristic time [19].
Extension, pause and stasis of the lamellipodia[Edit]
Extension of these newly formed branches occurs at the interface between the leading edge and the existing actin filament network [20] and is maintained by mechanisms such as actin treadmilling. As reviewed in [21] a number of models propose mechanisms by which actin filament dynamics lead to the generation of protrusive force, and subsequently lamellipodia extension. Early models, including the ‘Brownian ratchet’ theory [22] and the ‘Elastic ratchet’ theory [23], considered the process from a molecular level where each actin filament acted independently of others. Extending filaments would push against the membrane, and in the process a gap, produced either by membrane resistance or by the thermal wave like properties of an elastic non-rigid filament, would allow for the addition of G-actin onto the barbed end of extending filaments.
Although these models could account for the generation of protrusive force, they did not consider actin polymerization from an in vivoperspective where its dynamics are influenced by an array of factors. Direct interactions between growing actin filaments and the membrane were addressed in later models such as the ‘tethered ratchet’ model [24] where filaments were proposed to attach transiently to the membrane and protrusive force was generated by the addition of G-actin onto compressed filaments which had temporarily dissociated from the membrane. Similarly the influence of surface curvature was considered in the ‘elastic propulsion’ model [25] whilst the role of the greater filament network on filament polymerization was considered in the ‘autocatalytic branching’ theory [26].
With the specific factors that mediate force generation from F-actin assembly still to be defined, models such as those mentioned above will continue to be developed and built upon. It is clear however that actin treadmilling is integral to each model, being essential to both F-actin assembly and lamellipodia growth.
Filament extension occurs via the ‘actin treadmilling’ mechanism, with lamellipodial growth reflecting the balance between actin filament polymerization at the barbed ends and retrograde actin flow towards the cell body (reviewed in [27]). Motile cells adjust the rate of actin assembly in specific regions of the cell to outpace retrograde flow, thereby favoring cell contact and protrusion in a particular direction [28]. In certain motile cells (e.g. fish keratocytes), the actin cytoskeleton remains stationary relative to the substratum, indicating that there is virtually no retrograde actin flow and therefore the rate of actin polymerization equals the rate of protrusion [29].
Lamellipodial extension is often coupled to larger events that facilitate cell motility and mechanosensing. For example, extension of the actin filament network is essential in periodic contractions. This process, is primarily driven by myosin motor proteins and allows the cell to sense and measure the rigidity of its surroundings. Myosin motors localize behind the actin filament network, making filament extension essential to facilitate the rearward movement of older filaments towards these motors – a process resulting from the retrograde movement that occurs during filament extension.
Along with periodic contractions, lamellipodial extension may also occur along with peripheral membrane ruffling or with circular dorsal ruffles.
Although the lamellipodial actin network is highly dynamic, moments of pause and stasis have been reported [30]. This has been correlated to the angle of actin filaments at the membrane interface, with a greater number of ‘low angle’ filaments being seen to align in parallel to the leading edge during longer pauses [30]. These ‘low angle’ filaments continue to extend whilst shorter ‘high angle’ filaments depolymerize, resulting in a net reduction in protrusion [30]. Pause and stasis also allow time for maturation and elongation of nascent focal adhesions along the actin network [31]. This has been shown to occur particularly at the interface of lamellipodia and lamellae [31].
As the lamellipodia continues to spread and expand forward, adhesion sites form along the leading edge. These sites not only provide traction for the cells forward movement, but permit the cell to sense and measure the rigidity of its substrate. Formation of these adhesions is a complex process, involving a number of steps and functional modules. Particularly important in lamellipodia function is the spatial arrangement of adhesions at the leading edge. Lamellipodia extension and retraction is not uniform across the whole cell and similarly, adhesion sites will not be uniformly distributed. Instead a number of factors may influence arrangement including chemistry of the extracellular matrix, substrate stiffness and a cells growth conditions.
Interestingly, distribution of adhesion sites was found to be coupled to actin polymerization, with conformationally activated, but unliganded β1 integrins, shown to be interacting with elongating filaments [32]. Importantly, this interaction permits a sideways distribution along the leading edge, ensuring clusters of β1 integrin are positioned at the very front of cell protrusions. β1 integrin was also found, in the same study, to localize at the tips of growth cone filopodia [32]. Paxillin, a scaffold protein, has recently been shown to integrate physical cues sensed by adhesion complexes in order to confer directionality for lamellipodial protrusions [33].
Although these models could account for the generation of protrusive force, they did not consider actin polymerization from an in vivoperspective where its dynamics are influenced by an array of factors. Direct interactions between growing actin filaments and the membrane were addressed in later models such as the ‘tethered ratchet’ model [24] where filaments were proposed to attach transiently to the membrane and protrusive force was generated by the addition of G-actin onto compressed filaments which had temporarily dissociated from the membrane. Similarly the influence of surface curvature was considered in the ‘elastic propulsion’ model [25] whilst the role of the greater filament network on filament polymerization was considered in the ‘autocatalytic branching’ theory [26].
With the specific factors that mediate force generation from F-actin assembly still to be defined, models such as those mentioned above will continue to be developed and built upon. It is clear however that actin treadmilling is integral to each model, being essential to both F-actin assembly and lamellipodia growth.
Filament extension occurs via the ‘actin treadmilling’ mechanism, with lamellipodial growth reflecting the balance between actin filament polymerization at the barbed ends and retrograde actin flow towards the cell body (reviewed in [27]). Motile cells adjust the rate of actin assembly in specific regions of the cell to outpace retrograde flow, thereby favoring cell contact and protrusion in a particular direction [28]. In certain motile cells (e.g. fish keratocytes), the actin cytoskeleton remains stationary relative to the substratum, indicating that there is virtually no retrograde actin flow and therefore the rate of actin polymerization equals the rate of protrusion [29].
Lamellipodial extension is often coupled to larger events that facilitate cell motility and mechanosensing. For example, extension of the actin filament network is essential in periodic contractions. This process, is primarily driven by myosin motor proteins and allows the cell to sense and measure the rigidity of its surroundings. Myosin motors localize behind the actin filament network, making filament extension essential to facilitate the rearward movement of older filaments towards these motors – a process resulting from the retrograde movement that occurs during filament extension.
Along with periodic contractions, lamellipodial extension may also occur along with peripheral membrane ruffling or with circular dorsal ruffles.
Although the lamellipodial actin network is highly dynamic, moments of pause and stasis have been reported [30]. This has been correlated to the angle of actin filaments at the membrane interface, with a greater number of ‘low angle’ filaments being seen to align in parallel to the leading edge during longer pauses [30]. These ‘low angle’ filaments continue to extend whilst shorter ‘high angle’ filaments depolymerize, resulting in a net reduction in protrusion [30]. Pause and stasis also allow time for maturation and elongation of nascent focal adhesions along the actin network [31]. This has been shown to occur particularly at the interface of lamellipodia and lamellae [31].
As the lamellipodia continues to spread and expand forward, adhesion sites form along the leading edge. These sites not only provide traction for the cells forward movement, but permit the cell to sense and measure the rigidity of its substrate. Formation of these adhesions is a complex process, involving a number of steps and functional modules. Particularly important in lamellipodia function is the spatial arrangement of adhesions at the leading edge. Lamellipodia extension and retraction is not uniform across the whole cell and similarly, adhesion sites will not be uniformly distributed. Instead a number of factors may influence arrangement including chemistry of the extracellular matrix, substrate stiffness and a cells growth conditions.
Interestingly, distribution of adhesion sites was found to be coupled to actin polymerization, with conformationally activated, but unliganded β1 integrins, shown to be interacting with elongating filaments [32]. Importantly, this interaction permits a sideways distribution along the leading edge, ensuring clusters of β1 integrin are positioned at the very front of cell protrusions. β1 integrin was also found, in the same study, to localize at the tips of growth cone filopodia [32]. Paxillin, a scaffold protein, has recently been shown to integrate physical cues sensed by adhesion complexes in order to confer directionality for lamellipodial protrusions [33].
Focal adhesions in lamellipodia act as molecular clutches[Edit]
Once formed, focal adhesions essentially act as “molecular clutches”, promoting protrusion at the leading edge whilst suppressing membrane contraction (reviewed in [34, 35, 36]). Adhesions aid forward movement by regulating the forces produced by actin dynamics in different cellular compartments through several methods:
1) They aid membrane protrusion by resisting actin retrograde flow [5] and hence, indirectly promote the force produced by lamellipodial actin polymerization.
2) They convert myosin pulling forces at the lamellar interface into traction forces against the ECM that pulls the cell body forward [37, 38].
These two methods are interdependent and cooperatively contribute to the propulsive forces generated at the leading edge. The efficiency of the molecular clutch in converting this force into protrusion is variable. Because some components of the adhesion complex move along with the retrograde flow [39], the clutch slips [40]. The rate of forward protrusion increases however when actin and actin-adhesion linking components become more organized [41] and the retrograde flow of actin is subsequently slowed down [5, 42]. In this case the clutch can be described as “partially engaged”. As the adhesions grow and mature under stress, the clutch transforms from being “partially/locally engaged” to “engaged” [34] and hence can influence global cell behavior [43, 44].
Video: Focal adhesions are essential for cell spreading. Upon neuregulin treatment, cell-surface receptors Erb B3/B4 induce lamellipodia formation, most probably through activation of the Rac-WAVE-Arp2/3 pathway. During lamellipodial protrusion, numerous focal adhesions form along the cell periphery and can be visualized as fluorescent spots (GFP-VASP). [Source: Leticia Carramusa, Weizmann Institute of Science, Israel. Permission: Alexander Bershadsky, Mechanobiology Institute, Singapore.]
Experiments have demonstrated that a biphasic relationship exists between the rate of actin flow and traction stress [45]. Whilst they are inversely related in the lamellipodium where nascent adhesions are formed and actin flow rate is high, the relationship becomes linear in areas with larger adhesions and slow actin flow [5], generating maximal propulsion at intermediate flow rates [46].
Two recent studies propose stochastic models that explain the state of adhesion clutches in these two regimes [47, 48] (reviewed in [49]). One study describes how adhesion stability is modulated by the competition between energy required for maintaining the elastic bonds at the moving actin-clutch interface, and the energy dissipation that occurs when myosin pulls in the viscoelastic actin interior [47]. When the transmitted force and the speed of actin flow are moderate, much of the energy is invested in producing traction on the substrate. At this critical transition range, either an increase or decrease in the ratio of bound adhesion complexes could theoretically occur. In the second study however, based on known experimental results, it was assumed that at moderate actin flow rates adhesion clutches reach a quasi-equilibrium state between bound and unbound forms [48]. Thus the catch bond model of receptor-actin interactions are more convincing than the slip bond since it is more likely to lead to clutch engagement and adhesion growth, as observed in the experimental studies.
1) They aid membrane protrusion by resisting actin retrograde flow [5] and hence, indirectly promote the force produced by lamellipodial actin polymerization.
2) They convert myosin pulling forces at the lamellar interface into traction forces against the ECM that pulls the cell body forward [37, 38].
These two methods are interdependent and cooperatively contribute to the propulsive forces generated at the leading edge. The efficiency of the molecular clutch in converting this force into protrusion is variable. Because some components of the adhesion complex move along with the retrograde flow [39], the clutch slips [40]. The rate of forward protrusion increases however when actin and actin-adhesion linking components become more organized [41] and the retrograde flow of actin is subsequently slowed down [5, 42]. In this case the clutch can be described as “partially engaged”. As the adhesions grow and mature under stress, the clutch transforms from being “partially/locally engaged” to “engaged” [34] and hence can influence global cell behavior [43, 44].
Video: Focal adhesions are essential for cell spreading. Upon neuregulin treatment, cell-surface receptors Erb B3/B4 induce lamellipodia formation, most probably through activation of the Rac-WAVE-Arp2/3 pathway. During lamellipodial protrusion, numerous focal adhesions form along the cell periphery and can be visualized as fluorescent spots (GFP-VASP). [Source: Leticia Carramusa, Weizmann Institute of Science, Israel. Permission: Alexander Bershadsky, Mechanobiology Institute, Singapore.]
Experiments have demonstrated that a biphasic relationship exists between the rate of actin flow and traction stress [45]. Whilst they are inversely related in the lamellipodium where nascent adhesions are formed and actin flow rate is high, the relationship becomes linear in areas with larger adhesions and slow actin flow [5], generating maximal propulsion at intermediate flow rates [46].
Two recent studies propose stochastic models that explain the state of adhesion clutches in these two regimes [47, 48] (reviewed in [49]). One study describes how adhesion stability is modulated by the competition between energy required for maintaining the elastic bonds at the moving actin-clutch interface, and the energy dissipation that occurs when myosin pulls in the viscoelastic actin interior [47]. When the transmitted force and the speed of actin flow are moderate, much of the energy is invested in producing traction on the substrate. At this critical transition range, either an increase or decrease in the ratio of bound adhesion complexes could theoretically occur. In the second study however, based on known experimental results, it was assumed that at moderate actin flow rates adhesion clutches reach a quasi-equilibrium state between bound and unbound forms [48]. Thus the catch bond model of receptor-actin interactions are more convincing than the slip bond since it is more likely to lead to clutch engagement and adhesion growth, as observed in the experimental studies.
Force generation and cell translocation[Edit]
Interactions between actin filament networks and the focal adhesions to which they are linked results in the generation of forces. These forces may be exerted internally through actin bundle tension and filament network dynamics or externally as the cell pushes on its surroundings. A number of studies, discussed below, have focused on measuring the protrusive forces generated by lamellipodia [50, 51].
A study by Prass et al described the ‘whole cell stall force’ by placing an atomic force microscopy cantilever in the path of migrating keratocytes. This measurement represents the maximum force applied by the lamellipodia before the leading edge can no longer overcome the opposing force of the cantilever. At the moment this point is reached the process of cell body translocation stalls. In the case of migrating keratocytes, the maximal whole cell stall force was 40nN [50].
Importantly, observations from this study revealed that the force applied to the cantilever was generated by only a small region (3µM) of the lamellipodia, with the adjacent portions continuing to crawl forward, moving around the object. This not only highlights the dynamic nature of the actin cytoskeletal network within lamellipodia, but also illustrates that the total force is generated by independent filament components. In this case it was calculated that approximately 4pN of force was generated by each filament and with a whole cell stall force of 40nN, approximately 100 filaments were estimated to be generating the protrusive force per 1µM of the leading edge [50].
Protrusive forces have also been measured in non-migratory cells, with a higher resolution of detection through the use of optical tweezers. In one such study by Cojac et al, the forces exerted by lamellipodia during neuronal differentiation were compared to those produced byfilopodia and found to differ significantly. The forces produced by filopodia were consistently measured to be no larger than 2pN and were exerted for varying lengths of time of up to 15 seconds. in contrast the forces produced by lamellipodia ranged greatly from less than 1pN to more than 20pN – a force strong enough to displace trapped beads. Furthermore, these forces were exerted for an equally wide range of time, from less than 1 second to 30 seconds [51].
In another study conducted using optical tweezers, by Shahapure et al, the protrusive force of lamellipodia in rat dorsal ganglia (DRG) was measured, within a millisecond and picoNewton range [52]. In this case the maximal pressure exerted by DRG lamellipodia was ranged between 20 to 80pN/µm2. The ever-changing and complex nature of the generation of these forces was also noted. This was apparent from the alternating phases of rapid growth and retraction at the leading edge, further complicated by the influence of signals generated at adhesion sites. Adhesion sites in this case were in some cases associated with a transient inversion of lamellipodial velocity as well as transient retractions, all of which impinge on force generation [3].
Although each of the studies described above provide key quantitative insights into the total protrusive force generated by lamellipodia, it remains difficult to determine the specific contributions of the mechanisms driving these forces within each biological system. This is highlighted by the in vivo work of Prass et al, where numerous factors such as actin polymerization, the influence of motor proteins, local osmotic pressure and mechanical and chemical stimuli all influence lamellipodial protrusion [50].
The processes contributing to force generation during lamellipodial protrusion are varied in both their mechanisms and in their contribution. In some cases these processes will be redundant, whilst in other cases they will be crucial. Understanding which process to isolate, control or measure in an in vitro setting is a factor that currently limits our ability to properly define force generation. In line with this thinking, Shahapure et al recognized the fact that the dynamic nature of the lamellipodia causes the force it generates to be equally dynamic.
A study by Prass et al described the ‘whole cell stall force’ by placing an atomic force microscopy cantilever in the path of migrating keratocytes. This measurement represents the maximum force applied by the lamellipodia before the leading edge can no longer overcome the opposing force of the cantilever. At the moment this point is reached the process of cell body translocation stalls. In the case of migrating keratocytes, the maximal whole cell stall force was 40nN [50].
Importantly, observations from this study revealed that the force applied to the cantilever was generated by only a small region (3µM) of the lamellipodia, with the adjacent portions continuing to crawl forward, moving around the object. This not only highlights the dynamic nature of the actin cytoskeletal network within lamellipodia, but also illustrates that the total force is generated by independent filament components. In this case it was calculated that approximately 4pN of force was generated by each filament and with a whole cell stall force of 40nN, approximately 100 filaments were estimated to be generating the protrusive force per 1µM of the leading edge [50].
Protrusive forces have also been measured in non-migratory cells, with a higher resolution of detection through the use of optical tweezers. In one such study by Cojac et al, the forces exerted by lamellipodia during neuronal differentiation were compared to those produced byfilopodia and found to differ significantly. The forces produced by filopodia were consistently measured to be no larger than 2pN and were exerted for varying lengths of time of up to 15 seconds. in contrast the forces produced by lamellipodia ranged greatly from less than 1pN to more than 20pN – a force strong enough to displace trapped beads. Furthermore, these forces were exerted for an equally wide range of time, from less than 1 second to 30 seconds [51].
In another study conducted using optical tweezers, by Shahapure et al, the protrusive force of lamellipodia in rat dorsal ganglia (DRG) was measured, within a millisecond and picoNewton range [52]. In this case the maximal pressure exerted by DRG lamellipodia was ranged between 20 to 80pN/µm2. The ever-changing and complex nature of the generation of these forces was also noted. This was apparent from the alternating phases of rapid growth and retraction at the leading edge, further complicated by the influence of signals generated at adhesion sites. Adhesion sites in this case were in some cases associated with a transient inversion of lamellipodial velocity as well as transient retractions, all of which impinge on force generation [3].
Although each of the studies described above provide key quantitative insights into the total protrusive force generated by lamellipodia, it remains difficult to determine the specific contributions of the mechanisms driving these forces within each biological system. This is highlighted by the in vivo work of Prass et al, where numerous factors such as actin polymerization, the influence of motor proteins, local osmotic pressure and mechanical and chemical stimuli all influence lamellipodial protrusion [50].
The processes contributing to force generation during lamellipodial protrusion are varied in both their mechanisms and in their contribution. In some cases these processes will be redundant, whilst in other cases they will be crucial. Understanding which process to isolate, control or measure in an in vitro setting is a factor that currently limits our ability to properly define force generation. In line with this thinking, Shahapure et al recognized the fact that the dynamic nature of the lamellipodia causes the force it generates to be equally dynamic.
Focal Adhesion Disassembly and Retraction of the Trailing Edge[Edit]
Protrusion at the front and retraction at the rear are key force-generating processes at the cell periphery that culminate in the translocation of the cell. For forces to be translated into a net forward gain in cellular movement, the trailing edge must retract as the leading edge protrudes forward. In order for this to occur focal adhesions at the rear of the cell, and the actin filament network to which they are linked, must be disassembled. Prevention of this step would result in the cell being permanently anchored to its substrate.
Prior to retraction of the trailing edge, large-scale actomyosin activity at the rear of the cell reorganizes the cytoskeleton. This alters the actin flow and essentially polarizes the cell to influence the direction it will migrate [53]. The contractile forces generated by the actomyosin network facilitate the retraction of the trailing edge [54, 55] via a mechanism similar to that of Myosin Motors and Actin Filament Contractions. Myosin II is a major regulator of this contractility, generating increased tensile forces at the trailing edge and simultaneously relieving tension at the leading edge [56, 57]. This ultimately leads to retraction at the rear and extension at the front. Both myosin IIA and IIB are involved in this however each contributes differently. Myosin IIA is important in retraction and adhesion disassembly at the rear of the cell whilst myosin IIB plays a greater role in crosslinking of actin filaments and the establishment of a front-rear polarity [58, 59].
Focal adhesion disassembly eases separation of the trailing edge from the underlying substrate in response to the increase in actomyosin contractility. The subsequent retraction of the trailing edge results from the buildup of tensile forces at the rear of the cell which sever the interactions between the cytoskeleton and the cell substrate. These interactions are maintained by integrins however during retraction the integrins will remain attached to the substrate, whilst other components of the FA move towards the cell body and dissipate [60].
Further to the intracellular processes that promote detachment, namely FA disassembly and actomyosin contractility, extracellular events are also speculated to contribute (as reviewed in [61]). Extracellular matrix (ECM) proteases could digest the underlying substrate, whilst sheddases could cleave cell surface proteins bound to the substrate. Together these intracellular and extracellular events free the trailing edge of the cell from the substrate to which it is bound, allowing retraction to occur.
Prior to retraction of the trailing edge, large-scale actomyosin activity at the rear of the cell reorganizes the cytoskeleton. This alters the actin flow and essentially polarizes the cell to influence the direction it will migrate [53]. The contractile forces generated by the actomyosin network facilitate the retraction of the trailing edge [54, 55] via a mechanism similar to that of Myosin Motors and Actin Filament Contractions. Myosin II is a major regulator of this contractility, generating increased tensile forces at the trailing edge and simultaneously relieving tension at the leading edge [56, 57]. This ultimately leads to retraction at the rear and extension at the front. Both myosin IIA and IIB are involved in this however each contributes differently. Myosin IIA is important in retraction and adhesion disassembly at the rear of the cell whilst myosin IIB plays a greater role in crosslinking of actin filaments and the establishment of a front-rear polarity [58, 59].
Focal adhesion disassembly eases separation of the trailing edge from the underlying substrate in response to the increase in actomyosin contractility. The subsequent retraction of the trailing edge results from the buildup of tensile forces at the rear of the cell which sever the interactions between the cytoskeleton and the cell substrate. These interactions are maintained by integrins however during retraction the integrins will remain attached to the substrate, whilst other components of the FA move towards the cell body and dissipate [60].
Further to the intracellular processes that promote detachment, namely FA disassembly and actomyosin contractility, extracellular events are also speculated to contribute (as reviewed in [61]). Extracellular matrix (ECM) proteases could digest the underlying substrate, whilst sheddases could cleave cell surface proteins bound to the substrate. Together these intracellular and extracellular events free the trailing edge of the cell from the substrate to which it is bound, allowing retraction to occur.
Video: Retraction of the trailing edge of a motile cell. This video, on a repeated loop, shows the retraction of the rear of a motile cell. [Video uploaded to YouTube by UNCLineberger.]